Factors that cause migration
| Exploring for Oil and Gas Traps | |

| |
| Series | Treatise in Petroleum Geology |
|---|---|
| Part | Critical elements of the petroleum system |
| Chapter | Migration of petroleum |
| Author | Martin D. Matthews |
| Link | Web page |
| Store | AAPG Store |
Hydrocarbons migrate from a position of higher potential energy to one of lower potential energy. The spatial location of these energy differences defines the expected migration path. There are many sources for this energy that causes oil and gas migration.
Sources of energy
Three factors primarily cause hydrocarbon migration. All may be active at the same time during the migration process. Each factor produces energy from one or more sources. The table below lists these factors and their corresponding energy sources.
| Factors | Energy sources |
|---|---|
| Presence of oil or gas |
|
| Indirect effects on oil or gas due to burial |
|
| Decrease in pressure and temperature as a result of the upward migration of oil or gas |
|
Volume increase due to maturation
As maturation proceeds, solid kerogen is converted to liquid and gaseous hydrocarbons. When the activation energy levels of the kerogen are exceeded, this conversion is very rapid. The change in phase is accompanied by an increase in volume, preferentially over-pressuring the pore system within the source-rock unit and resulting in flow from the source-rock unit into the surrounding formations. This factor is of prime importance in source rocks.
Buoyancy
A free hydrocarbon phase rises in a water column because its density is less than that of water. This buoyancy force is proportional to the density difference and the height of the hydrocarbon column. It acts vertically. When the rising hydrocarbons encounter a low-permeability (high capillary entry pressure) sloping surface, they are deflected updip and the resultant force is decreased by an amount proportional to the slope of that surface.
Temperature and pressure effects on buoyancy
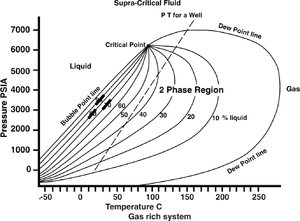
Rising temperature (T) increases the buoyancy force as the hydrocarbon is buried. The density of hydrocarbons decreases more rapidly than that of water as temperature increases. If the temperature is high enough, liquid hydrocarbons may alter to a gaseous phase. Rising pressure (P) decreases the buoyancy force as the hydrocarbons are buried. The density of hydrocarbons increases more rapidly than that of water as pressure increases. If the pressure is high enough, gaseous hydrocarbons may alter to a liquid phase.
Figure 1 summarizes the competing effects of pressure and temperature changes for a typical volatile oil.
Chemical potential
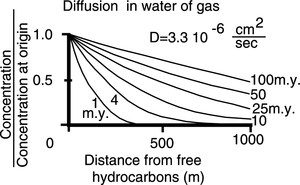
Diffusive forces spontaneously transfer hydrocarbons dissolved in water from areas of higher concentration to adjacent areas of lower concentration. The kilometers-long diffusion gradients of benzene and toluene within reservoirs demonstrate the effectiveness of this process. However, the lack of significant transport of benzene and toluene through barriers or seals indicates diffusion and active aqueous solution transport are minor mechanisms of the accumulation process. Figure 2 shows the diffusion of gas in water as a function of concentration at origin and distance from origin (free hydrocarbons).
Capillary imbibition
Capillary imbibition transfers interconnected free hydrocarbon phases from fine-grained to coarse-grained layers. This force is dominant within source rocks—especially at their contact with coarser beds. If the layer is internal to the source rock (such as a silty streak), it will store these hydrocarbons until a continuous hydrocarbon network connects it with an external coarse-grained layer. If the coarse-grained layer is external, thick, and laterally extensive, it will act as a carrier bed and postexpulsion migration will begin. Capillary imbibition exceeds buoyancy as the force responsible for transferring the hydrocarbon phase to the carrier bed, resulting in downward as well as upward charging from source rocks.
Sediment compaction
Burial results in the downward motion of each sediment package and is accompanied by a decrease in porosity as it compacts. The resultant motion of water is continually upward with respect to the sediment. The water motion with respect to the sediment/water interface, however, is downward because some water is continually trapped as sedimentation continues. In order for compaction-driven fluids to escape the sediment/water interface, they must move laterally into areas of concentrated upward flow.