Hydrology and sandstone diagenesis
| Exploring for Oil and Gas Traps | |

| |
| Series | Treatise in Petroleum Geology |
|---|---|
| Part | Predicting the occurrence of oil and gas traps |
| Chapter | Predicting reservoir system quality and performance |
| Author | Dan J. Hartmann, Edward A. Beaumont |
| Link | Web page |
| Store | AAPG Store |
Type of water flushes
Much diagenesis occurs in open chemical systems whose initial chemistry is set at deposition. After that, the chemistry of the system changes as flowing water moves chemical components through pores and causes either leaching or cementation of grains. Diffusion also moves chemicals in and out of rocks, although at significantly lower rates. During deep burial, chemical systems close and diagenesis is primarily by pressure solution and quartz overgrowths.[3]
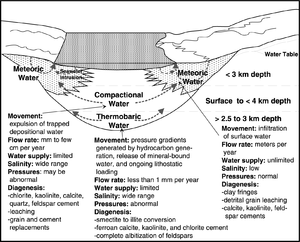
Galloway[1] lists three types of flow of water in a basin:
- Meteoric flow—water infiltrates shallow portions of a basin from precipitation or surface waters. Deeper infiltration occurs from (a) eustatic sea level changes and/or (b) tectonic elevation of basin margins.
- Compactional flow—compaction expels water upward and outward from the pores of sediments.
- Thermobaric flow—water moves in response to pressure gradients caused by generation of hydrocarbons, release of mineral-bound water, and/or increased heat flow.
Figure 1 shows the water movement processes mentioned above.
Pore-water chemistry
Depositional environment and climate control initial pore-water chemistry of a sandstone. When the rock is buried below the level of meteoric groundwater influence, pore-water chemistry changes as a result of two things:
- Increasing mineral solubility due to increasing temperatures.
- Acidic fluids released by maturing organic-rich shales or organic matter in sandstone. Acidic pore water leaches carbonate cement and grains.
Eh-pH graph
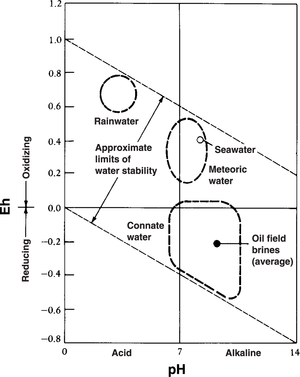
Figure 2 is an Eh–pH diagram, showing the approximate distribution of various types of subsurface fluids.
Pore-water chemistry and cements
The table below lists common sandstone cements and the water chemistry associated with precipitation.
| Cement | pH | Water type | Typical cement derivation |
|---|---|---|---|
| Quartz | Acidic | Meteoric feldspars | Clay compaction, weathering of Ca-Na |
| Calcite | Alkaline | Marine | Dissolution of skeletal material and carbonate minerals; precipitation from bicarbonate and calcium in sea water |
| Kaolinite | Acid | Meteoric | Breakdown of K-feldspar or mica grains by fresh water |
| Illite | Alkaline | Marine | Conversion of smectite to illite, hydrolysis of micas, intense weathering of K-feldspar |
| Chlorite | Alkaline | Brackish | Weathering of basic volcanic detritus and Fe-Mg minerals |
| Smectite | Alkaline | Brackish to marine | Breakdown of plagioclase grains and mafic minerals rich in Ca and Na |
Subsurface dissolved solids
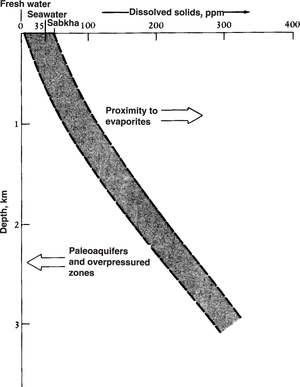
Figure 3 shows the general trend of increasing dissolved solids in subsurface fluids with increasing depth.
See also
- Sandstone porosity and permeability prediction
- Sandstone diagenetic processes
- Effect of composition and texture on sandstone diagenesis
- Influence of depositional environment on sandstone diagenesis
- Predicting sandstone reservoir porosity
- Predicting sandstone permeability from texture
- Estimating sandstone permeability from cuttings
References
- ↑ 1.0 1.1 Galloway, W. E., 1984, Hydrogeologic regimes of sandstone diagenesis, in D. A. McDonald, and R. C. Surdam, eds., Clastic Diagenesis: AAPG Memoir 37, p. 3–14.
- ↑ Harrison, W. J., and R. N. Tempel, 1993, Diagenetic pathways in sedimentary basins, in A. D. Horbury, and A. G. Robinson, eds., Diagenesis and Basin Development: AAPG Studies in Geology 36, p. 69–86.
- ↑ Wilson, M. D., 1994a, Non-compositional controls on diagenetic processes, in M. D. Wilson, ed., Reservoir Quality Assessment and Prediction in Clastic Rocks: SEPM Short Course 30, p. 183–208. Discusses the effect that variables such as temperature and pressure have on diagenesis of sandstones. A good reference for predicting sandstone reservoir system quality.
- ↑ 4.0 4.1 Shelley, R. C., 1985, Elements of Petroleum Geology: San Francisco, W.H. Freeman, 449 p.